In the market we have at our disposal several types of batteries. A battery is capable of managing energy to supply it and thus allow certain devices and appliances to function. Lithium-ion batteries are the powerhouse of much of the modern world, from smartphones, laptop tablets to electric cars and airplanes. Every scientific advance that improves battery performance is relevant, and these are the biggest battery innovations of 2021.
Some advancements come from experimenting with alternative materials while others come from the approach of completely redesigning a device and how it works from scratch. These are some of the most creative and interesting examples.

Towards a faster charge
One of the ways scientists are working to improve the charging rate of batteries is by using porous structures for the anode, one of its two electrons. This offers a greater contact area with the liquid electrolyte that carries the lithium ions and allows them to diffuse more easily through the material. Consequently, the batteries charge much faster.
In November, scientists at the University of Twente created an anode with a material called nickel niobate. It has an open and regular crystalline structure with repetition of the same channels that facilitate the transport of ions. Worked on a full battery cell, scientists found it offered ultra-fast recharge rates , up to 10 times faster than current ion batteries. The researchers note that nickel niobate has a higher bulk density than graphite, which could also lead to lighter and more compact commercial batteries.
Reviving lithium
Scientists at Stanford University have discovered a way to neutralize harmful groups of “dead lithium” and revive them to increase the performance of a battery. The team of scientists discovered that “adding a high current voltage during recharging stimulated the action of this inactive lithium, causing it to slip ‘like a worm’ and reconnect with the electrode, increasing the life of the electrode. the battery by 30% ”.
A breakthrough that could bring improvements in the design of fast-charging batteries or rechargeable batteries. This problem is real in next-generation lithium metal batteries , whose potential is to hold up to 10 times more energy. Therefore, this advance could bring with it new very promising solutions.
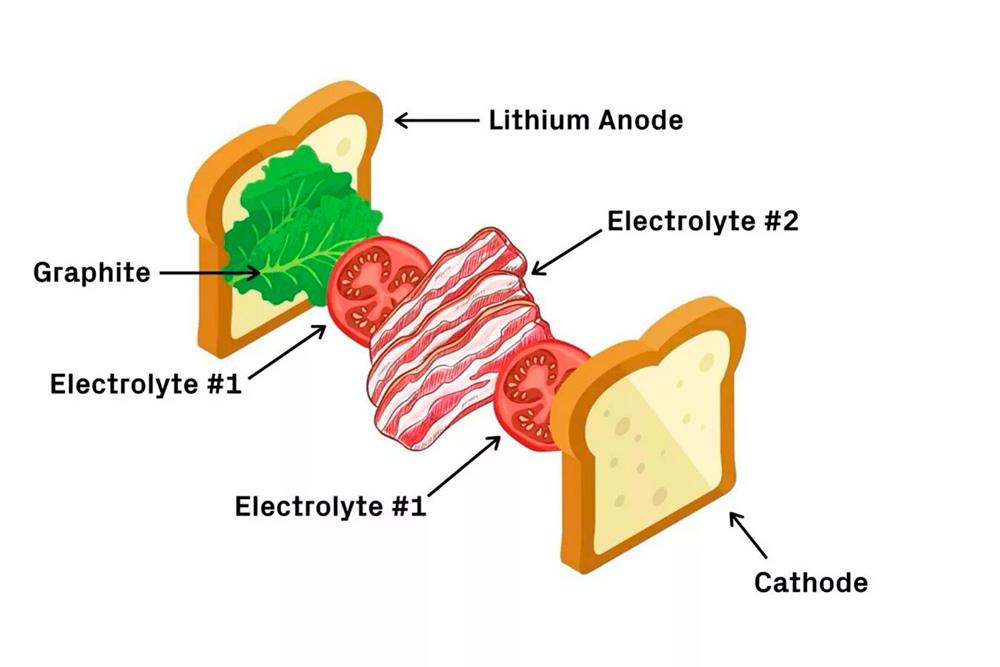
New sandwich style battery
Lithium metal has a much higher capacity and energy density than graphite and copper. This is why scientists see so much potential in lithium metal batteries.
Harvard materials scientist Xin Li unveiled a new sandwich-style battery in May that may be able to overcome some of the stability issues that are plaguing lithium metal designs thus far. After testing, the scientific team was able to verify that the battery retained 82% of its capacity after 10,000 charge cycles. Most promising, it demonstrated a type of current density that could one day allow electric vehicles to charge in 20 minutes.
Nature as a source of inspiration
In October, a team of scientists from the United States looked at another interesting solution to the stability problems associated with lithium metal batteries. This time, they turned to nature for inspiration. This breakthrough, once again, started from the notion of using a solid electrolyte rather than a liquid one to carry charge. In this case the scientists used cellulose nanofibrils derived from wood .
“These microscopic polymer tubes combined with copper to form a solid ion conductor, with tiny openings between the polymer chains that acted as ‘ion superhighways,’ allowing the lithium ions to travel with record efficiency.” This means that the material has a conductivity between 10 and 100 times higher than that of other polymer ion conductors.
New take on an old design
Chlorine alkali metal batteries have been around since the 1970s and offer high energy density. Highly reactive chlorine only lasts one use. In August, scientists at Stanford University came up with a way to stabilize this reaction and allow these types of batteries to recharge.
The solution consisted of an electrode material “made of porous carbon that absorbed erratic chlorine molecules and safely converted them to sodium chloride, their original form before discharge.” A cycle that could be repeated up to 200 times in an experimental battery that offered around six times the density of current lithium-ion technology.
Very thin strips of lithium
Scientists at the US Department of Energy’s Pacific Northwest National Laboratory (PNNL) “focused on what is known as the solid electrolyte interface (SEI) , which is a thin film on top of the anode that it plays an important control role by controlling which molecules enter from the electrolyte during the cycle ”.
They found a novel solution in the form of very thin strips of lithium with a width of around 20 microns, much thinner than human hair. The team’s bag cell battery prototype featured this anode that retained 76% of its capacity for 600 cycles.
Use of solid electrolyte
This advance features a battery with a “semi-solid” electrode made of sodium and potassium alloys that researchers compare to the material dentists use to fill cavities, due to its firmness and ability to flow and mold.
“When this material comes into contact with solid electrolyte, it has just the right amount of elasticity to avoid the kind of cracks that would form in a stiffer and more brittle electrode material.” This material allowed for higher current densities , around 20 times more, paving the way for much higher charge speeds.